Puma Biotechnology Announces FDA Advisory Committee to Review Neratinib for the Extended Adjuvant Treatment of HER2-Positive Ear
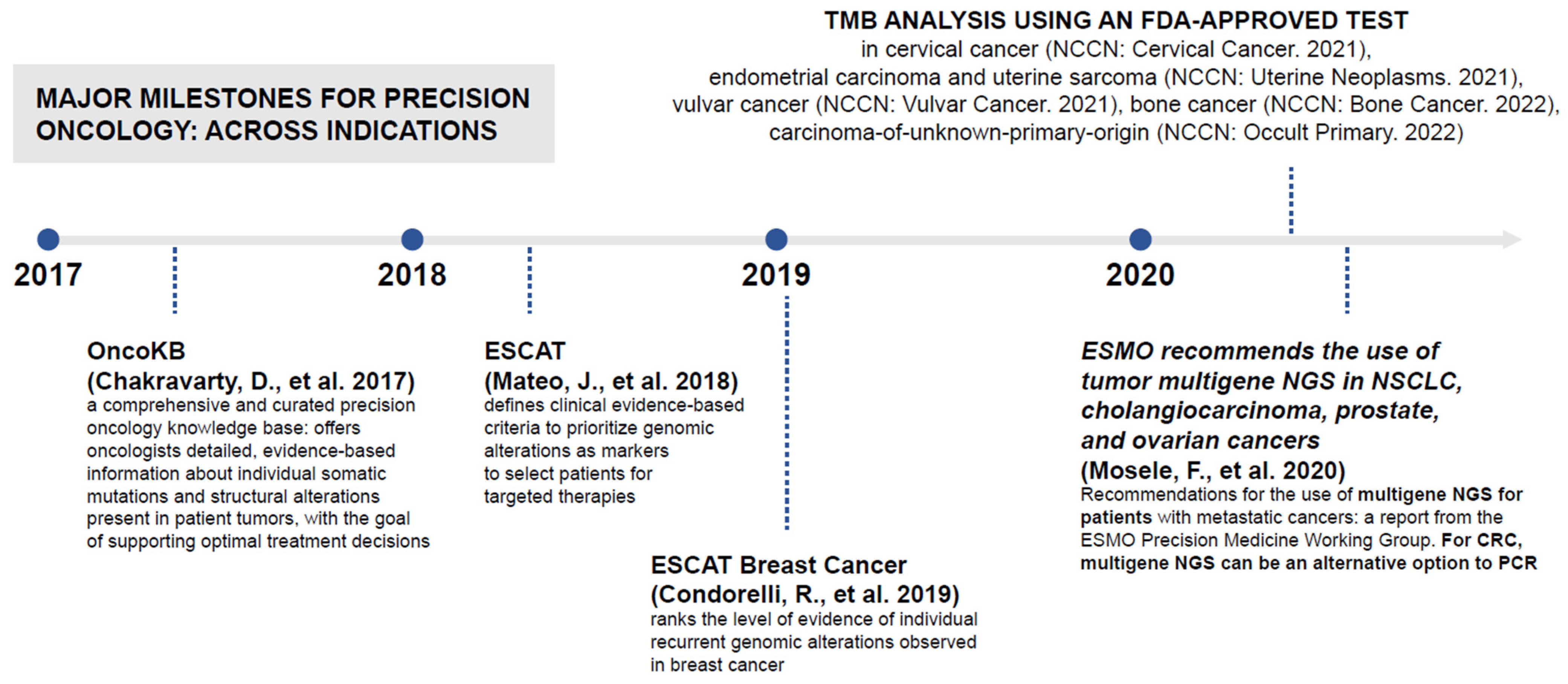
JPM | Free Full-Text | Identifying the Steps Required to Effectively Implement Next-Generation Sequencing in Oncology at a National Level in Europe

Restoring PUMA induction overcomes KRAS-mediated resistance to anti-EGFR antibodies in colorectal cancer | Oncogene






















