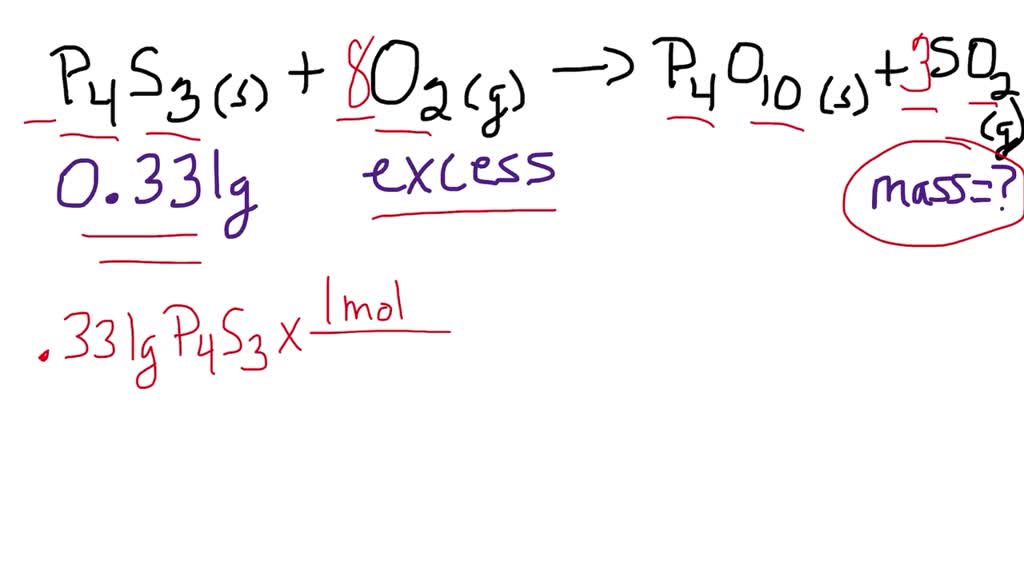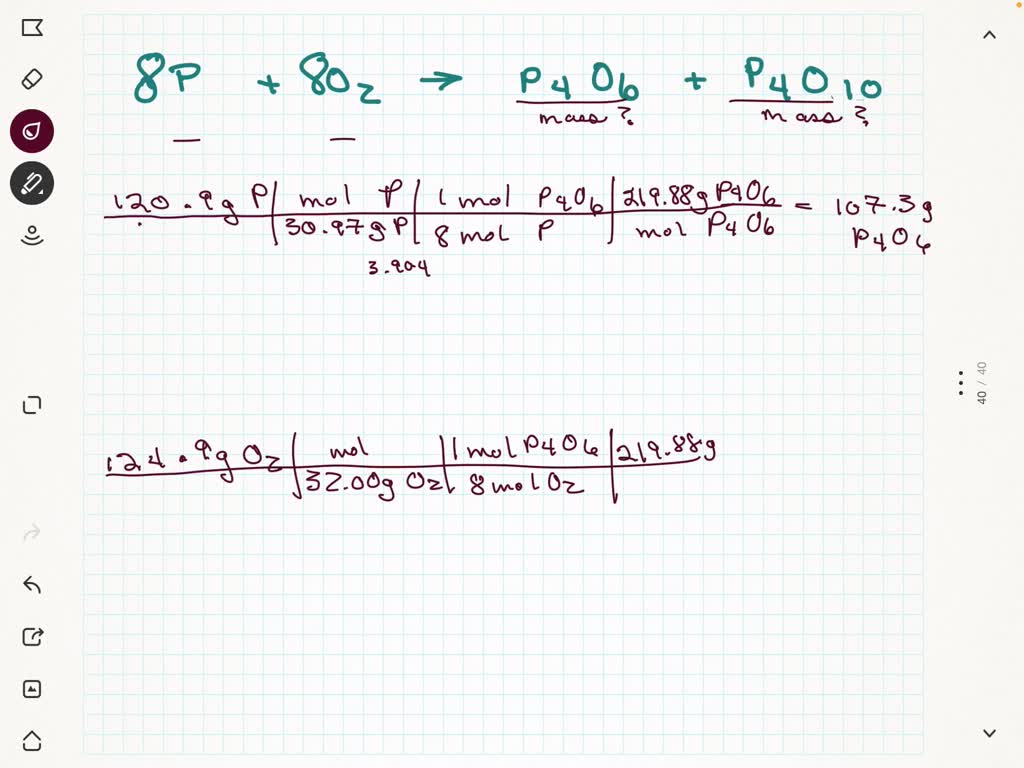
SOLVED: A mixture of 120.9 g of P and 124.9 g of O2 reacts completely to form P4O6 and P4O10. Find the masses of P4O6 and P4O10 that are formed by the reaction.
11 The mass of P4O10 produce if 440 gm of P4S is mixed with 384 gm of O2 Is given, P4S3 + O2 — > P4O10 + SO2 OPTIONS >> 568gm, 426gm , 284gm, 396gm.
a) What is the limiting reactant when 0.200 mol of P4 and 0.200mol of O2 react according to: P4 + 5O2 = P4O10 (b) Calculate the percent yield if 10.0 g of