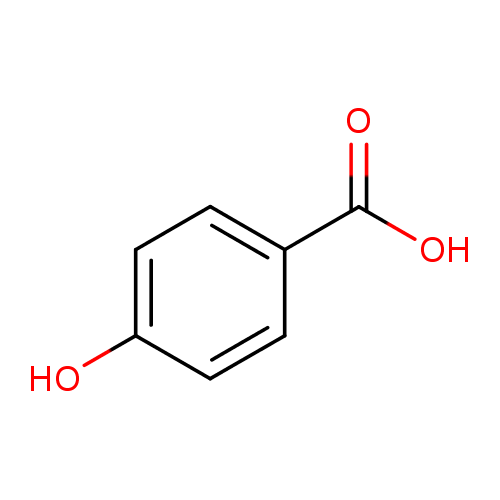
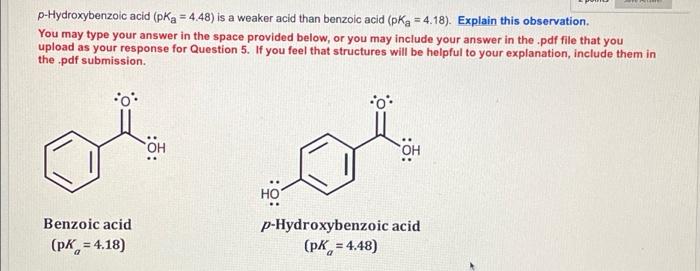
Reaction pathway of the degradation of the p-hydroxybenzoic acid by sulfate radical generated by ionizing radiations - ScienceDirect

Deprotonation of p-Hydroxybenzoic Acid: Does Electrospray Ionization Sample Solution or Gas-Phase Structures? | Journal of the American Chemical Society

A Reliable and Efficient First Principles-Based Method for Predicting pKa Values. 2. Organic Acids | The Journal of Physical Chemistry A
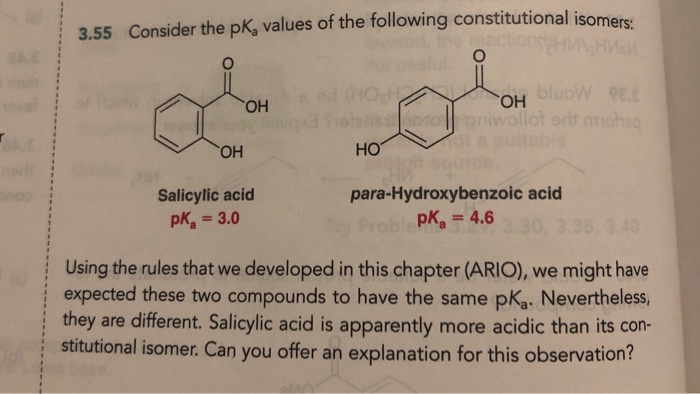
Does cyanide show the ortho effect in benzoic acid? If not, then what are the exceptions of the ortho effect in benzoic acid? - Quora
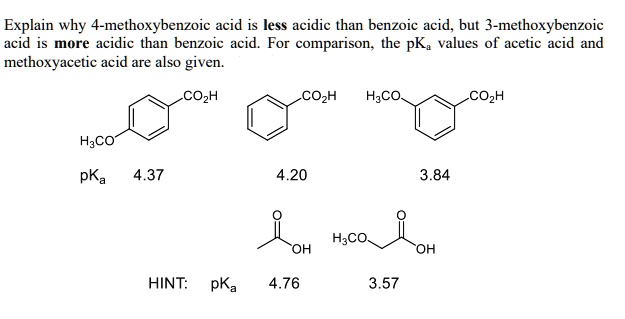
SOLVED: Explain why 4-methoxybenzoic acid is less acidic than benzoic acid, but 3-methoxybenzoic acid is more acidic than benzoic acid. For comparison, the pK values of acetic acid and methoxyacetic acid are

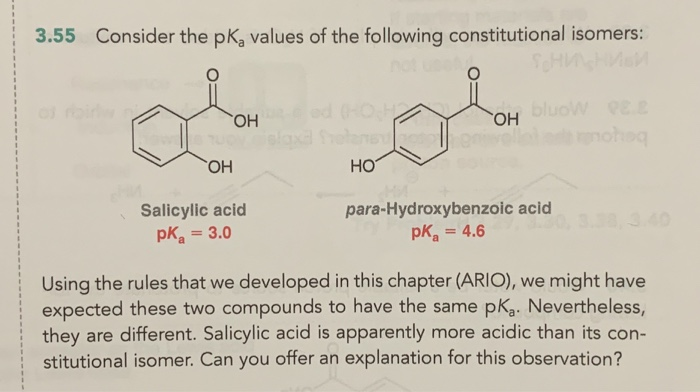
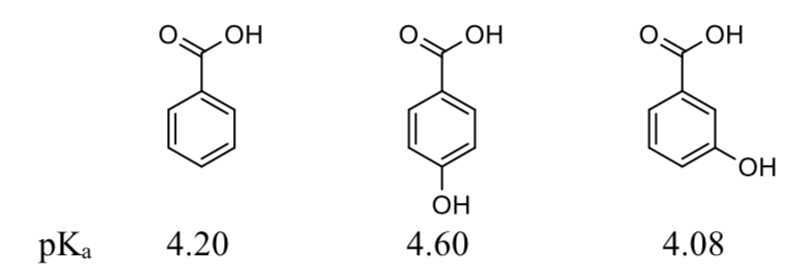
Calculated and experimental pK a values of ortho-substituted benzoic... | Download Scientific Diagram