Effect of time on p-cresol oxidation. Reaction conditions: p-cresol... | Download Scientific Diagram

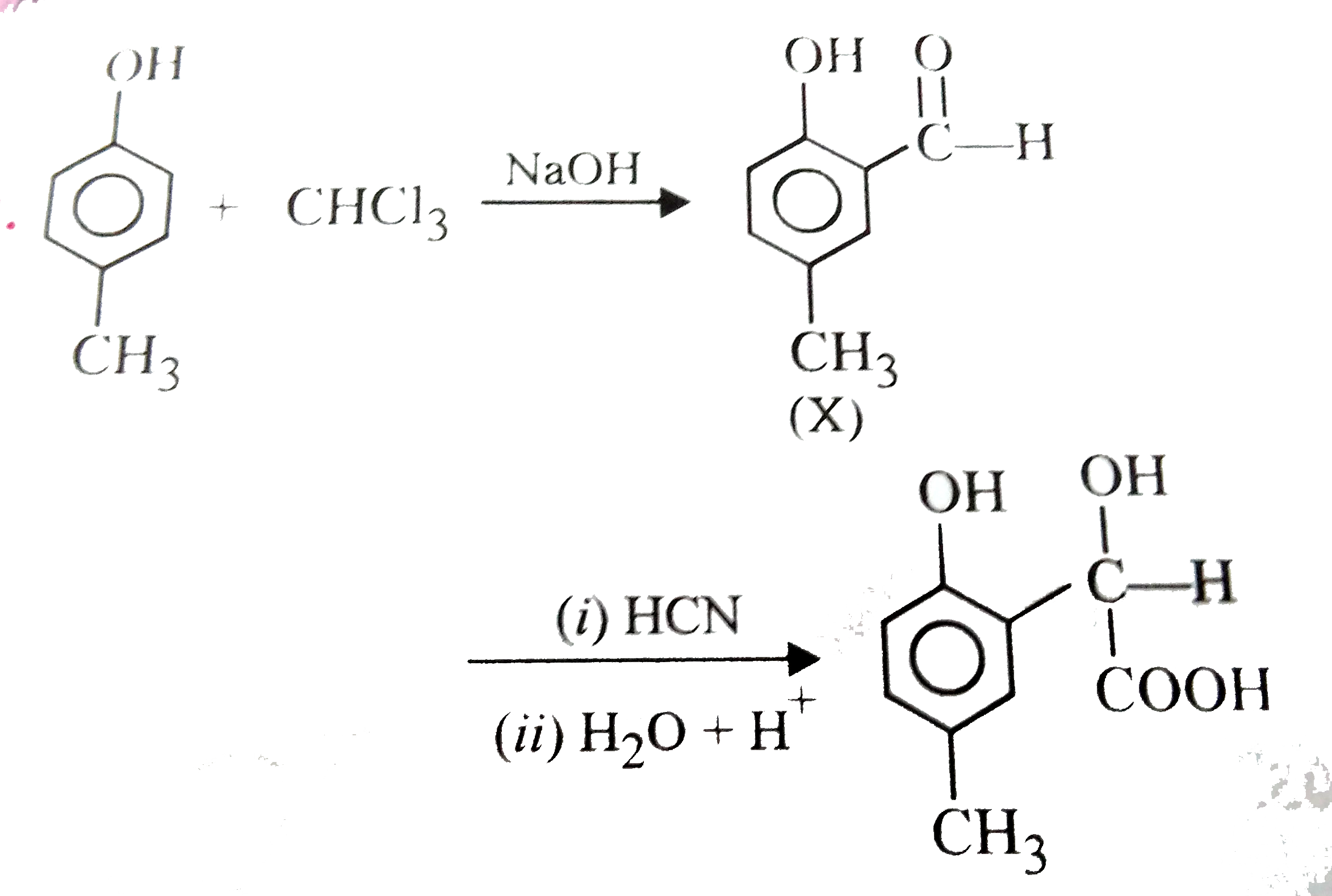
p - Cresol reacts with chloroform in alkaline medium to give the compound A which adds hydrogen cyanide to form the compound B. The latter on acidic hydrolysis gives chiral carboxylic acid .
![PDF] Autoxidation ofp-cresol top-hydroxy benzaldehyde using CoCl2, CoAPO-5 and CoAPO-11 | Semantic Scholar PDF] Autoxidation ofp-cresol top-hydroxy benzaldehyde using CoCl2, CoAPO-5 and CoAPO-11 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8e845603d2d4dcfb4c60dc392c455393d6a9d577/2-Figure1-1.png)
PDF] Autoxidation ofp-cresol top-hydroxy benzaldehyde using CoCl2, CoAPO-5 and CoAPO-11 | Semantic Scholar
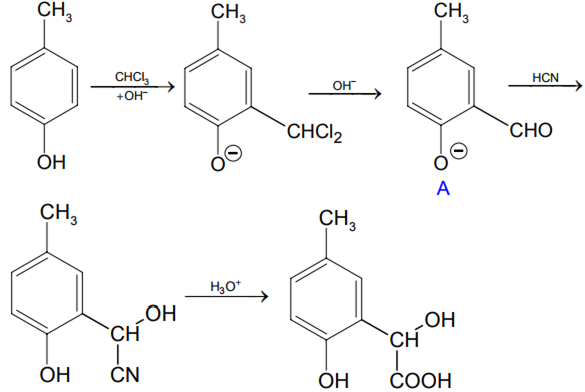
p-cresol reacts with chloroform in the alkaline medium to give the compound A which adds hydrogen cyanide to form, the compound B. The latter on acidic hydrolysis gives chiral carboxylic acid. The

Effect of time on p-cresol oxidation. Reaction conditions: p-cresol... | Download Scientific Diagram
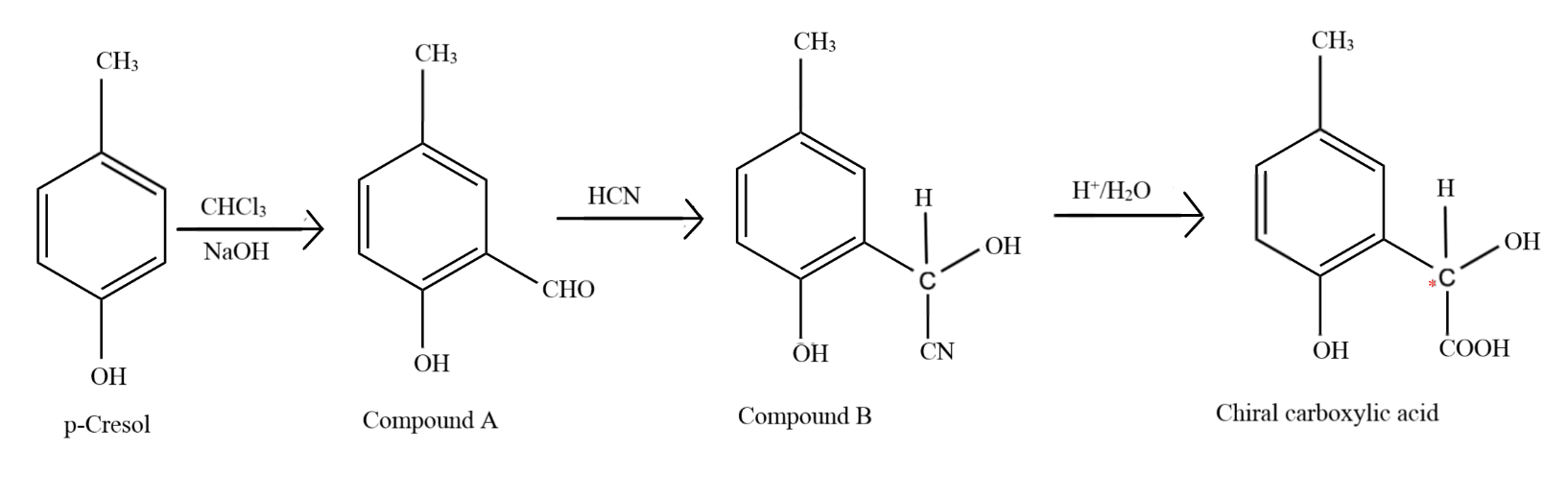
p-Cresol reacts with chloroform in alkaline medium to give the compound A which adds hydrogen cyanide to form the compound B. The latter on acidic hydrolysis gives chiral carboxylic acid. The structure

A Fully Continuous-Flow Process for the Synthesis of p-Cresol: Impurity Analysis and Process Optimization | Organic Process Research & Development

organic chemistry - Products of Reimer–Tiemann reaction of 4-methylphenol - Chemistry Stack Exchange

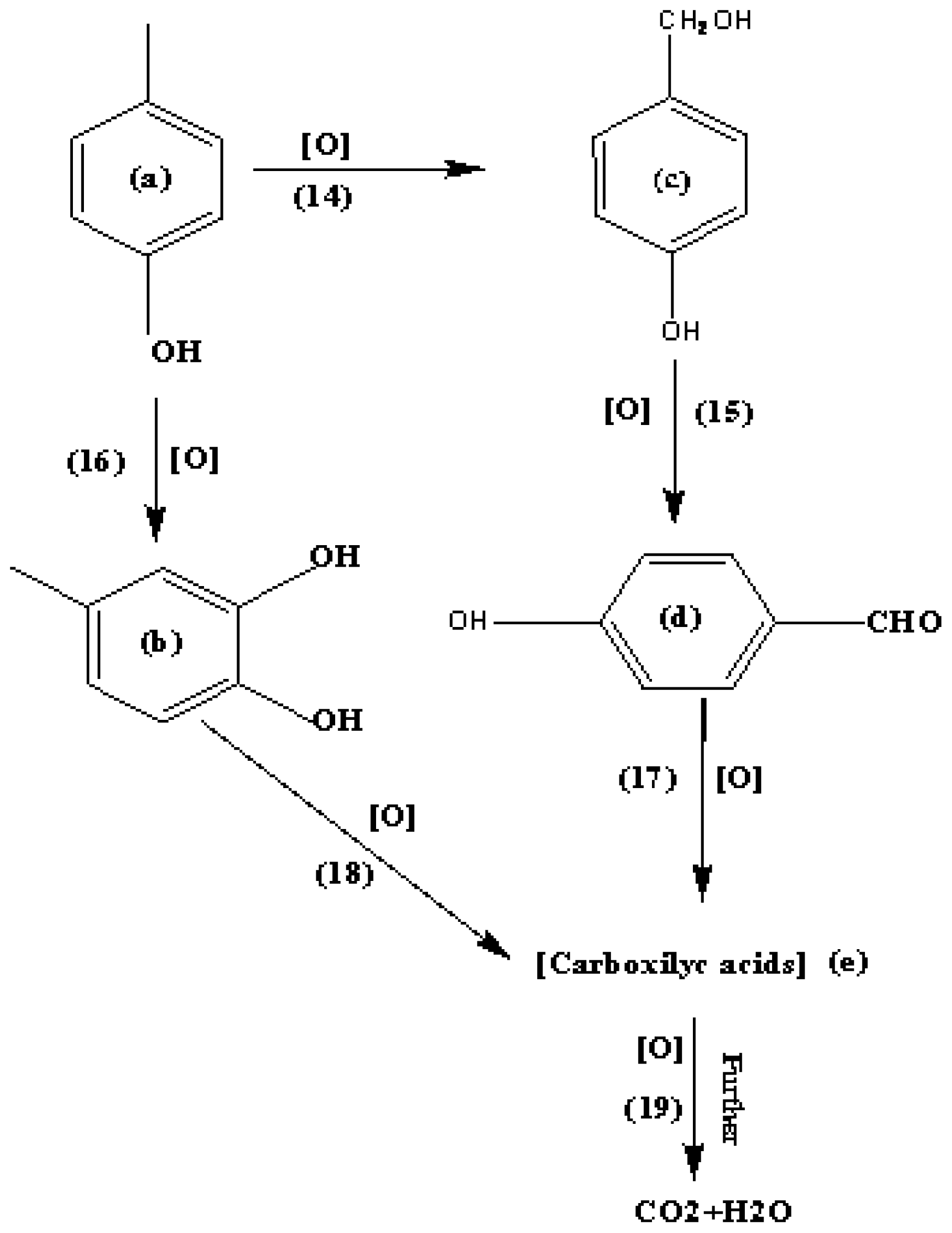
Reaction kinetics of liquid phase air oxidation of p-cresol to p-hydroxybenzaldehyde - ScienceDirect

P- cresol react with chloroform in alkaline medium to give compound `(A)` which adds `HCN` to for - YouTube

SOLVED: Question14 Explain the following: p-cresol is one of the products during the preparation of p-iodotoluene from toluidine Which of the following statement is true Not Yet answered Maked out of 1

Selective Synthesis of p-Hydroxybenzaldehyde by Liquid-Phase Catalytic Oxidation of p-Cresol | Organic Process Research & Development

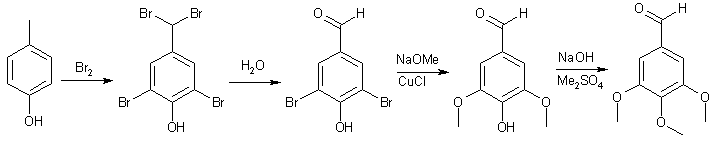
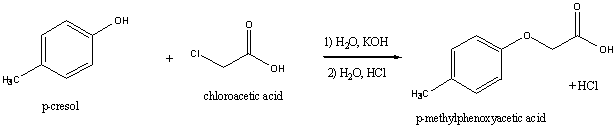
![Synthesis of 3,4,5-Trimethoxybenzaldehyde from p-Cresol - [www.rhodium.ws] Synthesis of 3,4,5-Trimethoxybenzaldehyde from p-Cresol - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/pictures/tmba-2.gif)
![PDF] A Mathematical Model for the Benzylation of p -Cresol with Benzyl Alcohol | Semantic Scholar PDF] A Mathematical Model for the Benzylation of p -Cresol with Benzyl Alcohol | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4aa7fe64552b832c2cac544d8aa08b30216bbcc8/6-TableV-1.png)

![Synthesis of 3,4,5-Trimethoxybenzaldehyde from p-Cresol - [www.rhodium.ws] Synthesis of 3,4,5-Trimethoxybenzaldehyde from p-Cresol - [www.rhodium.ws]](https://erowid.org/archive/rhodium/chemistry/pictures/tmba-1.gif)