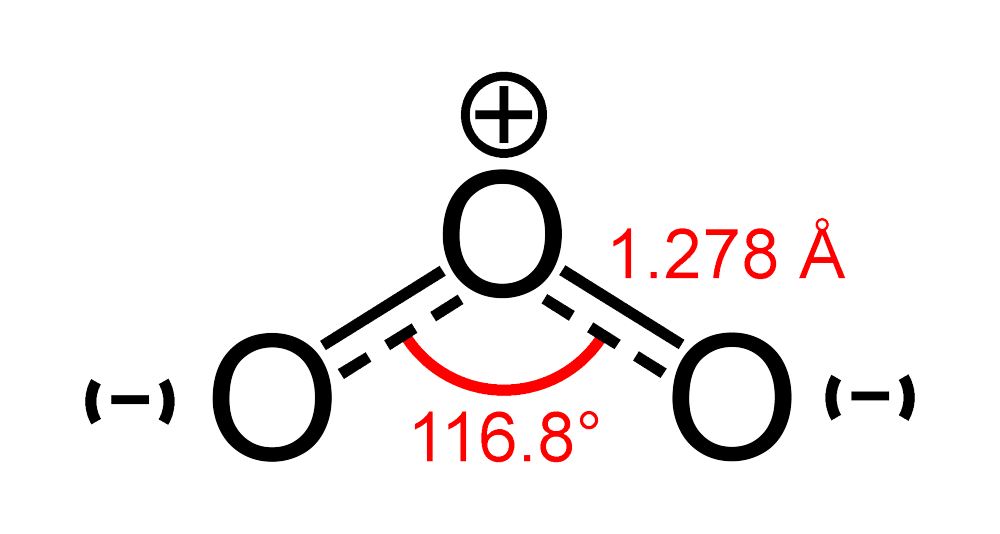
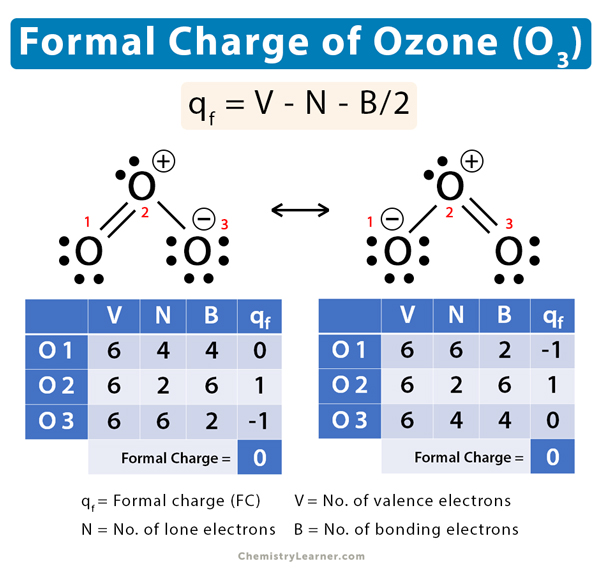
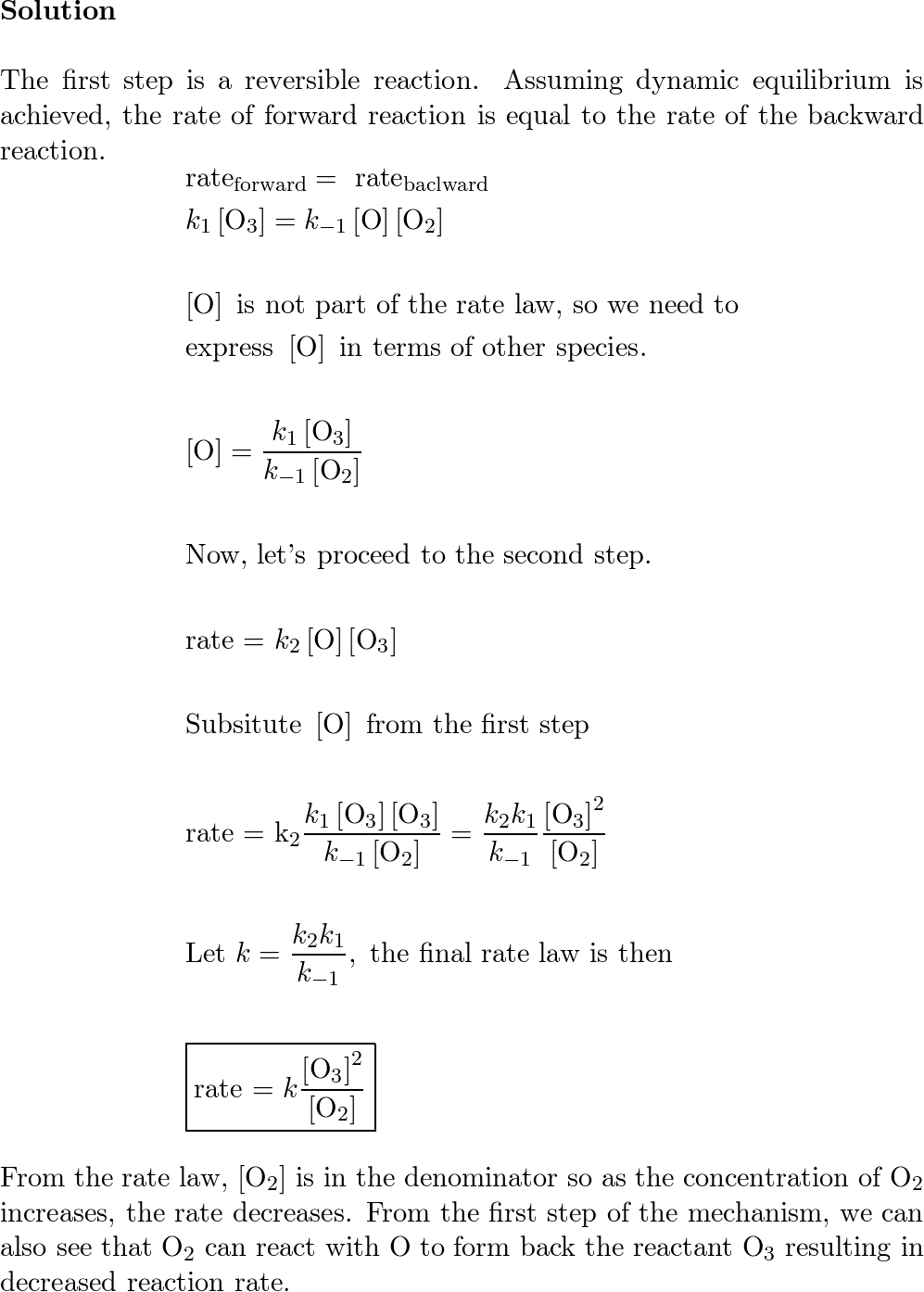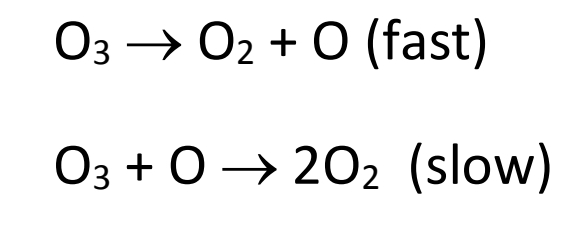On Rates and Mechanisms of OH and O3 Reactions with Isoprene-Derived Hydroxy Nitrates | The Journal of Physical Chemistry A

Amazon.com: Enerzen O-UVC3 - HEPA + UV Light + 40,000 mg/h Industrial Ozone Generator for Eliminating Odors - O3 Machine Air Ionizer Deodorizer for Home, Room, Smoke, Car, Pet : Home & Kitchen
![The ozone, O3, of the stratosphere can be decomposed by reaction with nitrogen oxide (commonly called nitric oxide),NO, from high-flying jet aircraft. The rate expression is rate = k[O3][NO]. Which of the The ozone, O3, of the stratosphere can be decomposed by reaction with nitrogen oxide (commonly called nitric oxide),NO, from high-flying jet aircraft. The rate expression is rate = k[O3][NO]. Which of the](https://homework.study.com/cimages/multimages/16/7133470169922890101680733.png)
The ozone, O3, of the stratosphere can be decomposed by reaction with nitrogen oxide (commonly called nitric oxide),NO, from high-flying jet aircraft. The rate expression is rate = k[O3][NO]. Which of the

Amazon.com: Enerzen O-922D - Digital Ozone Generator for Eliminating Odors - O3 Machine Air Ionizer Deodorizer with Adjustable Settings for Any Room Size : Home & Kitchen

SOLVED: A possible mechanism for the decomposition of Ozone is: O3 = 02 + 0 with forward and reverse rate constants k1 and k1 0 + O3 + 2 02 with rate
![For the reaction O3(g) + O(g) → 2O2(g) , if the rate law expression is rate = k[O3][O] , the molecularity and order of the reaction respectively are: For the reaction O3(g) + O(g) → 2O2(g) , if the rate law expression is rate = k[O3][O] , the molecularity and order of the reaction respectively are:](https://d1hhj0t1vdqi7c.cloudfront.net/v1/TDZKcEFiaXlCcFE=/sd/)
For the reaction O3(g) + O(g) → 2O2(g) , if the rate law expression is rate = k[O3][O] , the molecularity and order of the reaction respectively are:

Ozone, O3, Trioxygen, Inorganic Molecule. it is an Allotrope of Oxygen Stock Vector - Illustration of dioxygen, molecule: 197174422
![The chemical reaction, 2O2⟶3O2 proceeds as: O3 O2 + [O] (fast) + O3⟶2O2 (slow) The rate law expression will be: The chemical reaction, 2O2⟶3O2 proceeds as: O3 O2 + [O] (fast) + O3⟶2O2 (slow) The rate law expression will be:](https://dwes9vv9u0550.cloudfront.net/images/6173668/3a9b6913-3fa9-46cd-9c10-14063b8916de.jpg)
The chemical reaction, 2O2⟶3O2 proceeds as: O3 O2 + [O] (fast) + O3⟶2O2 (slow) The rate law expression will be:

SOLVED: A suggested mechanism for the decomposition of ozone is: O3 0z + 0 (fast) 02 + 0 O3 (fast) 0+O3 202 (slow) What is the rate law for this reaction from