Cr(VI) Reduction by Microsecond Pin-to-Pin Discharges Generated in an Aqueous Solution | SpringerLink
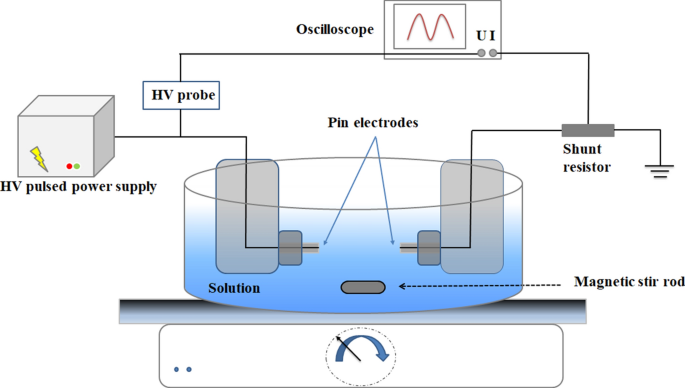
Cr(VI) Reduction by Microsecond Pin-to-Pin Discharges Generated in an Aqueous Solution | SpringerLink

Palladium Oxidase Catalysis: Selective Oxidation of Organic Chemicals by Direct Dioxygen‐Coupled Turnover - Stahl - 2004 - Angewandte Chemie International Edition - Wiley Online Library
Papers Past | Parliamentary Papers | Appendix to the Journals of the House of Representatives | 1913 Session I | EXPENDITURE OF THE PUBLIC ACCOUNT (STATEMENT IN...

Examining the Scope of Deriving β-Aryl Enones from Enol Silanes as Ketone Equivalents via Pd(II)-Mediated Sequential Dehydrosilylation and Arylation | The Journal of Organic Chemistry