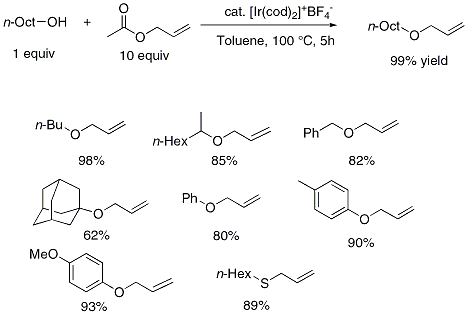
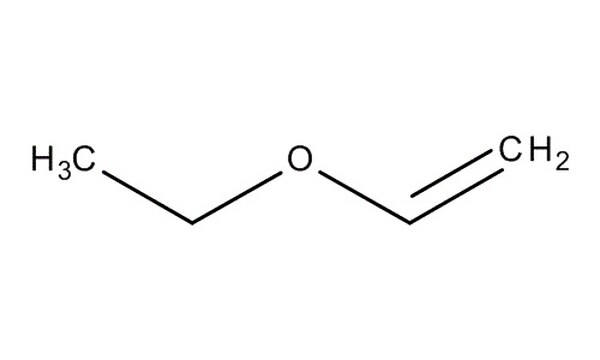
Explain why methyl vinyl ether (CH2=CHOCH3) is not a reactive dienophile in the Diels-Alder reaction. | Homework.Study.com
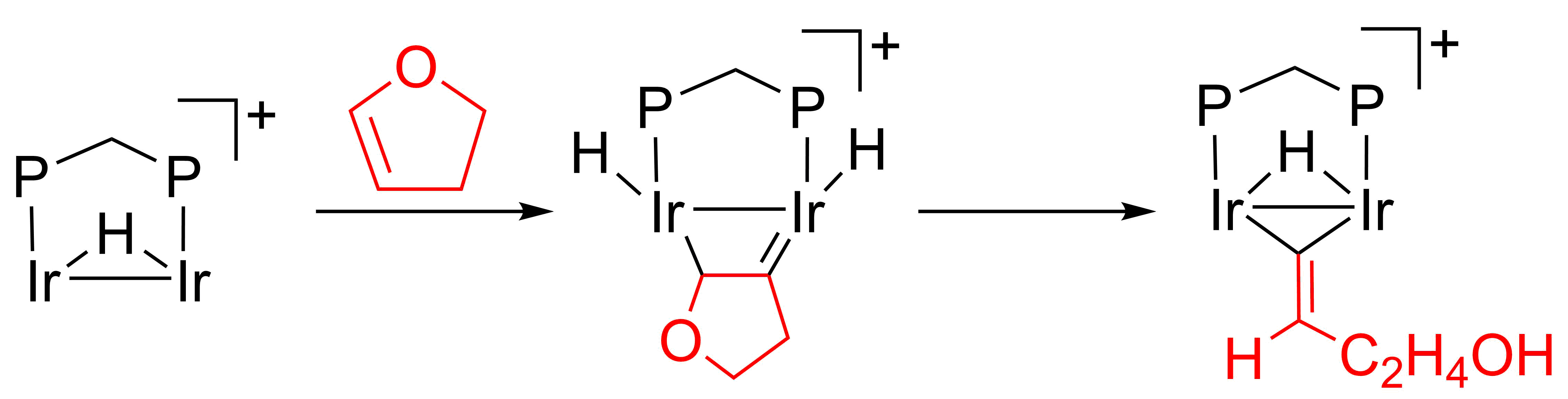
Inorganics | Free Full-Text | Successive Activation of C–H and C–O Bonds of Vinyl Ethers by a Diphosphine and Hydrido-Bridged Diiridium Complex

Living cationic polymerization of α‐methyl vinyl ethers using SnCl4 - Yonezumi - 2008 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library
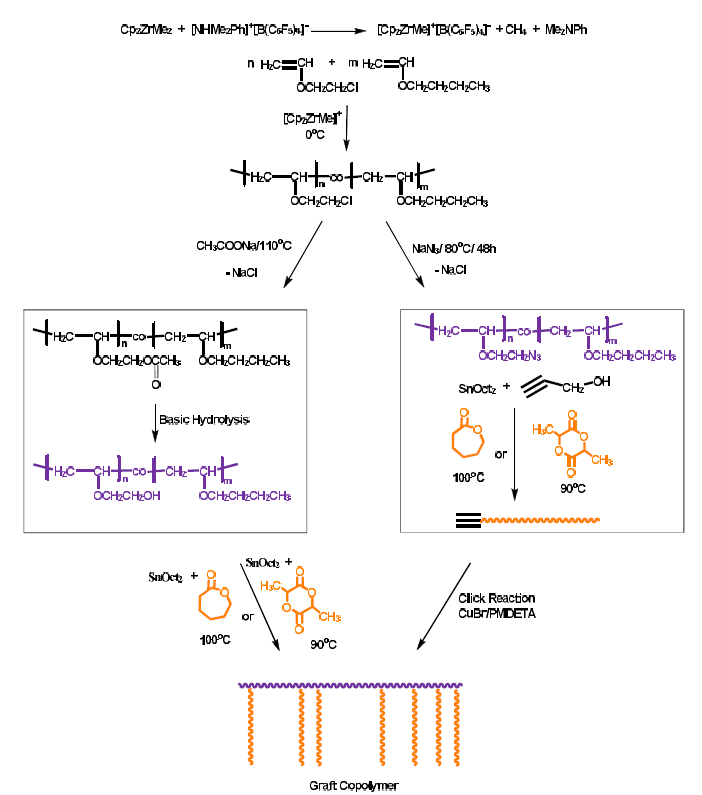
Polymers | Free Full-Text | Statistical Copolymers of n-Butyl Vinyl Ether and 2-Chloroethyl Vinyl Ether via Metallocene-Mediated Cationic Polymerization. A Scaffold for the Synthesis of Graft Copolymers

Synthesis of poly(vinyl ether) plastics for optical use by cationic copolymerization of tricyclodecyl vinyl ether with n‐butyl vinyl ether - Namikoshi - 2007 - Journal of Polymer Science Part A: Polymer Chemistry -

Recent advances in applications of vinyl ether monomers for precise synthesis of custom-tailored polymers - ScienceDirect

Pd-Catalyzed Three-Component Reaction of Anilines, Ethyl Vinyl Ether, and Nitro-Paraffin: Assembly of β-Nitroamines | Organic Letters

When methyl vinyl ether reacts with a strong acid, the proton adds to C2 exclusively, instead of C1 or the oxygen atom. Draw the three protonated forms of methyl vinyl ether and
![Synthonix, Inc > benzyl vinyl ether - [B26636] Synthonix, Inc > benzyl vinyl ether - [B26636]](https://synthonix.com/images/product/26636.png)











![POLY[TETRAFLUOROETHYLENE-CO-PERFLUORO (ALKYL VINYL ETHER)] | 26655-00-5 POLY[TETRAFLUOROETHYLENE-CO-PERFLUORO (ALKYL VINYL ETHER)] | 26655-00-5](https://www.chemicalbook.com/CAS/20180808/GIF/26655-00-5.gif)