Using the data presented in the pressure-versus-volume drawing below (x = 1.00E+4 Pa and y = 2.00E-3 m^3), calculate the magnitude of the work done when the system changes from A to
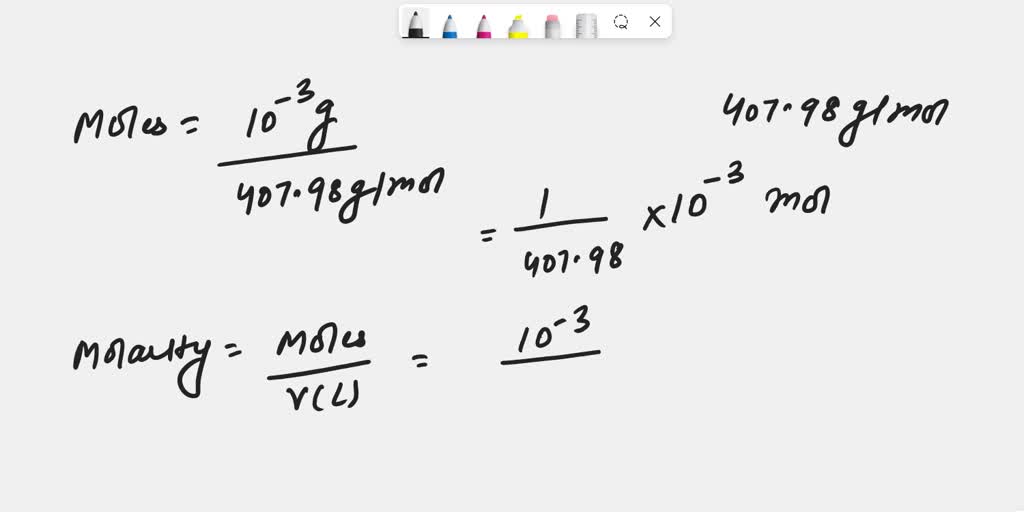
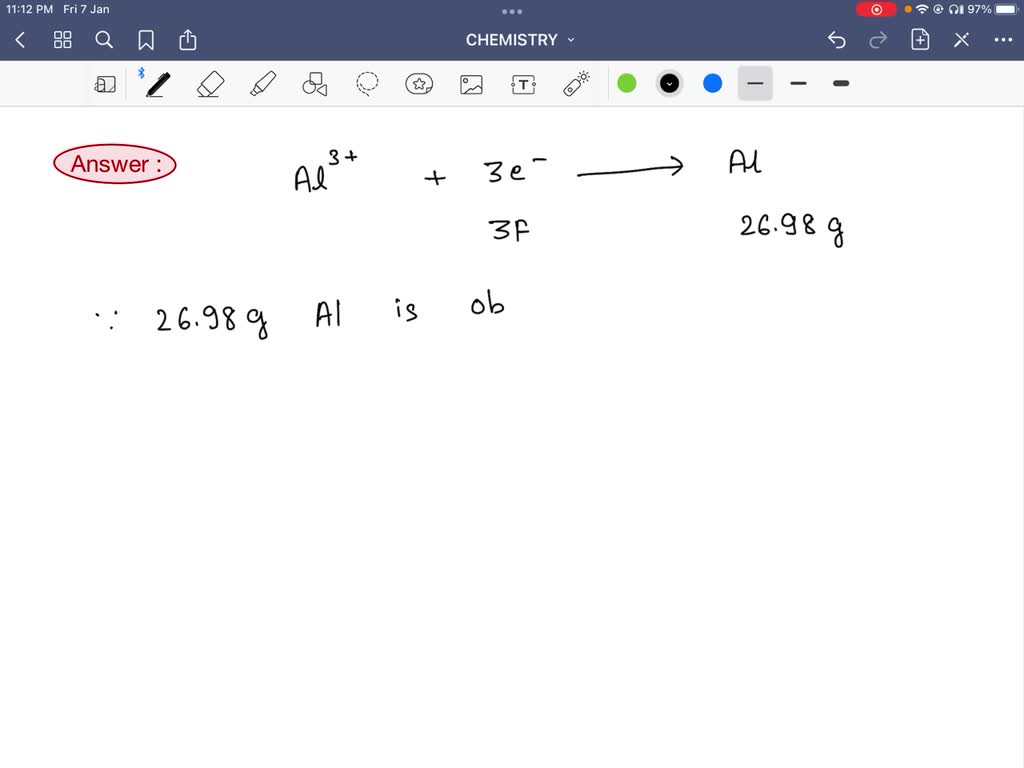
SOLVED: 1. Calculate the Molarity of 1.00E-3% mass crystal violet solution assuming at this concentration the density of the solution is 1.00 g/mL and the molar mass of crystal violet is 407.98

Min Choi on Twitter: "3. Support for Larger AI Models With current pace of AI model growth in both size and complexity, they require powerful infrastructure that can scale to meet these

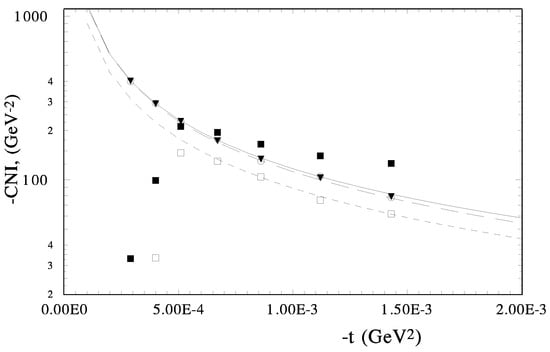
Distribution Coefficient Model for Zirconium and Technetium Extraction from Nitric Acid Solution | Industrial & Engineering Chemistry Research

Using the data presented in the pressure-versus-volume drawing below (x = 1.00E+4 Pa and y = 2.00E-3 m^3), calculate the magnitude of the work done when the system changes from A to
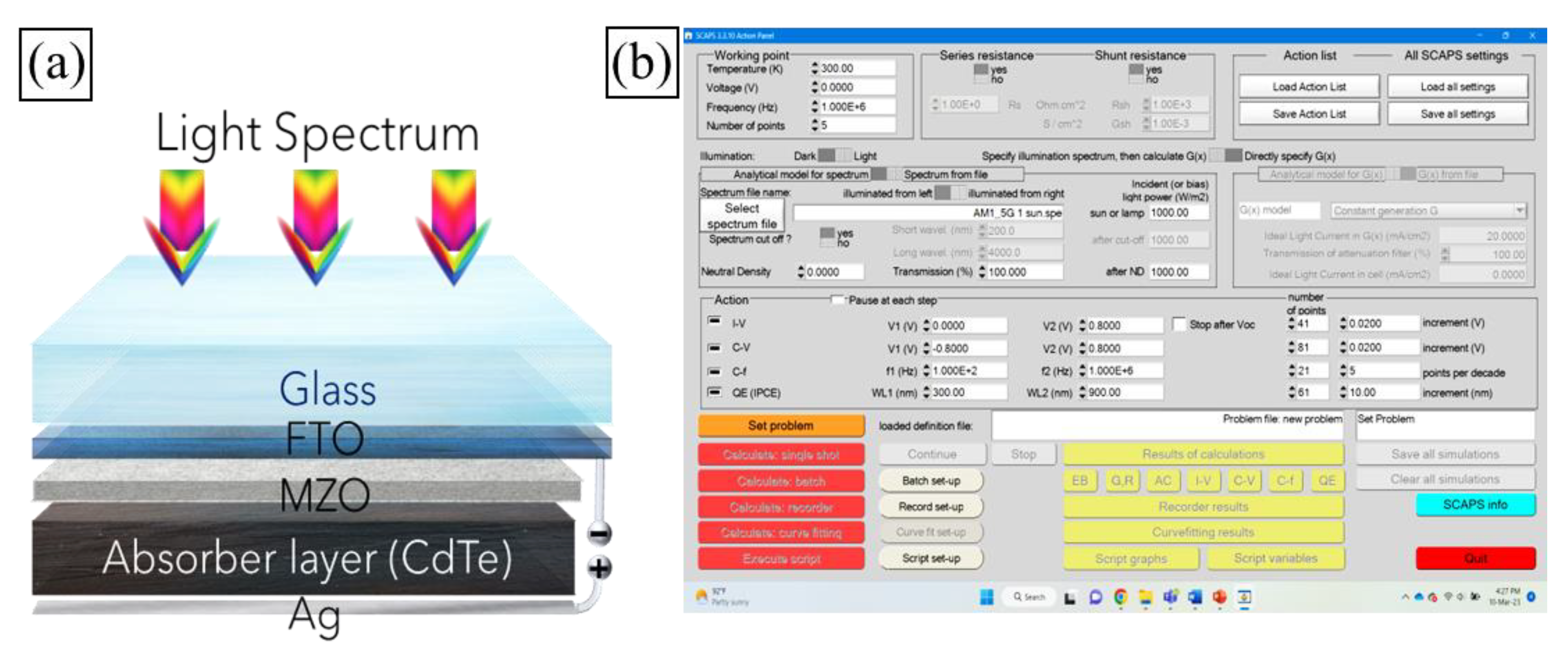
Crystals | Free Full-Text | Effect of Cu2Te Back Surface Interfacial Layer on Cadmium Telluride Thin Film Solar Cell Performance from Numerical Analysis

Evaluation and modelling of UPM pulp mill wastewater treatment plant in Fray Bentos, Uruguay - Master Theses Open Access - Repository IHE Delft Institute for Water Education

![Solved Time (sec) 0 1600 3200 [A] 1.00E-2 M 5.04E-3 M | Chegg.com Solved Time (sec) 0 1600 3200 [A] 1.00E-2 M 5.04E-3 M | Chegg.com](https://media.cheggcdn.com/study/81d/81dbc152-9c7e-4974-8ada-7171675bcdc3/image)

![駿河屋 -<中古>1.00-E-0008[R]:[煌めくステージへ]丸山彩(アニメ・ゲーム) 駿河屋 -<中古>1.00-E-0008[R]:[煌めくステージへ]丸山彩(アニメ・ゲーム)](https://www.suruga-ya.jp/database/pics_light/game/gg829326.jpg)